Medication Administration
Properly Recording NDC Information with Medication Administration
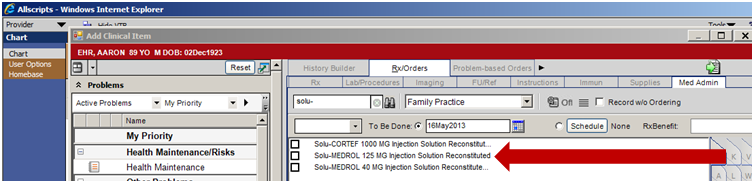
1. Search the ACI, under the Med Admin tab, for the medication in question. This example will utilize Solu-MEDROL 125 MG.
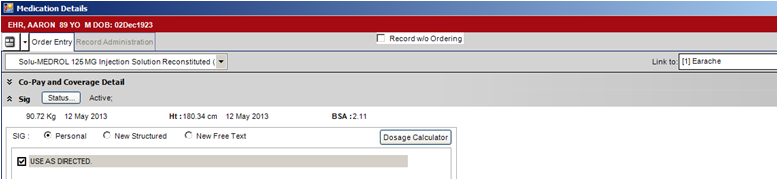
2. Properly complete all fields on the Order Entry tab for the medication.
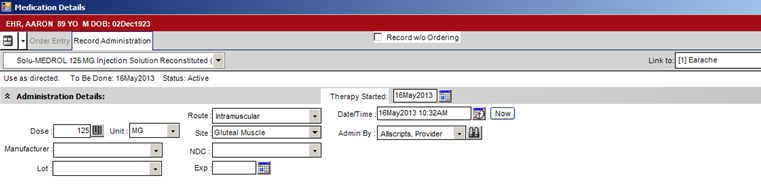
3. Click on the Record Administration tab and fill in all required fields. These are typically Dose, Unit, Route, Site, Manufacturer, NDC, Lot and Expiration Date, but may vary by organization.
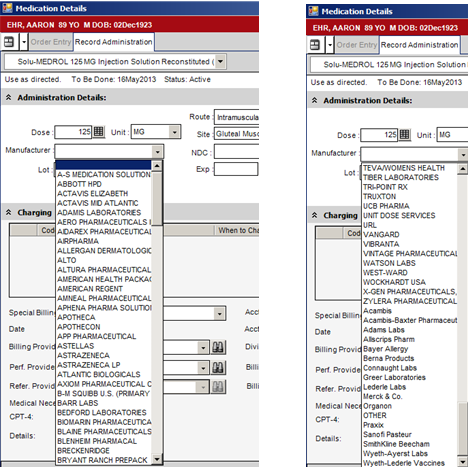
4. When selecting the Manufacturer, utilize the pull-down menu as seen below. 99% of the time, the correct entry will be found in ALL CAPS at the top of the list. NOTE – if you don’t see the entry you are looking for, verify you have not confused the Distributor with the Manufacturer.
5. If you select an entry listed in ALL CAPS, such as the A-S MEDICATION SOLUTIONS below, the available NDC codes for that manufacturer will auto-populate in that pull-down.
6. If you select one of entries not listed in ALL CAPS, no entries will be populated in the NDC.
7. One other common mistake is to select a related medication such as Solu-MEDROL 125 MG seen above versus MethylPREDNISolone Sodium Succ 125 MG seen below. The Manufacturer listings may or may not be the same, but the NDC codes will not mirror one another.
8. Be sure you are documenting the medication in the EHR that you are actually administering and you are selecting the manufacturer, not the distributor, and you will have a much higher chance of success appropriately documenting Medication Administrations in the practice.